- Drug discovery
METPlatform
- Are there more effective ways to find drugs working against brain metastasis?
A clinically-compatible drug-screening platform based on organotypic cultures identifies vulnerabilities to prevent and treat brain metastasis.
Zhu L, et al. EMBO Molecular Medicine. (2022).
Stratification of radiosensitive brain metastases based on an actionable S100A9/RAGE resistance mechanism.
Monteiro C, Miarka L, et al. Nature Medicine (2022).
Protocol to generate murine organotypic brain cultures for drug screening and evaluation of anti-metastatic efficacy
Lucía Zhu, Lauritz Miarka, Patricia Baena, María Perea-García, Manuel Valiente. STAR Protocols (2023)
Patients with brain metastasis face a dismal prognosis partly because of the limited therapeutic strategies available. Historically, the poor prognosis and the lack of information regarding the access of drugs to the brain were enough to exclude these patients from clinical trials.
Obviously, this fact has not contributed to challenge the status of brain metastasis as an unmet clinical need.
We realized that strategies that could score many drugs being applied to a preparation that recapitulates clinically-relevant aspects of metastases might be useful.
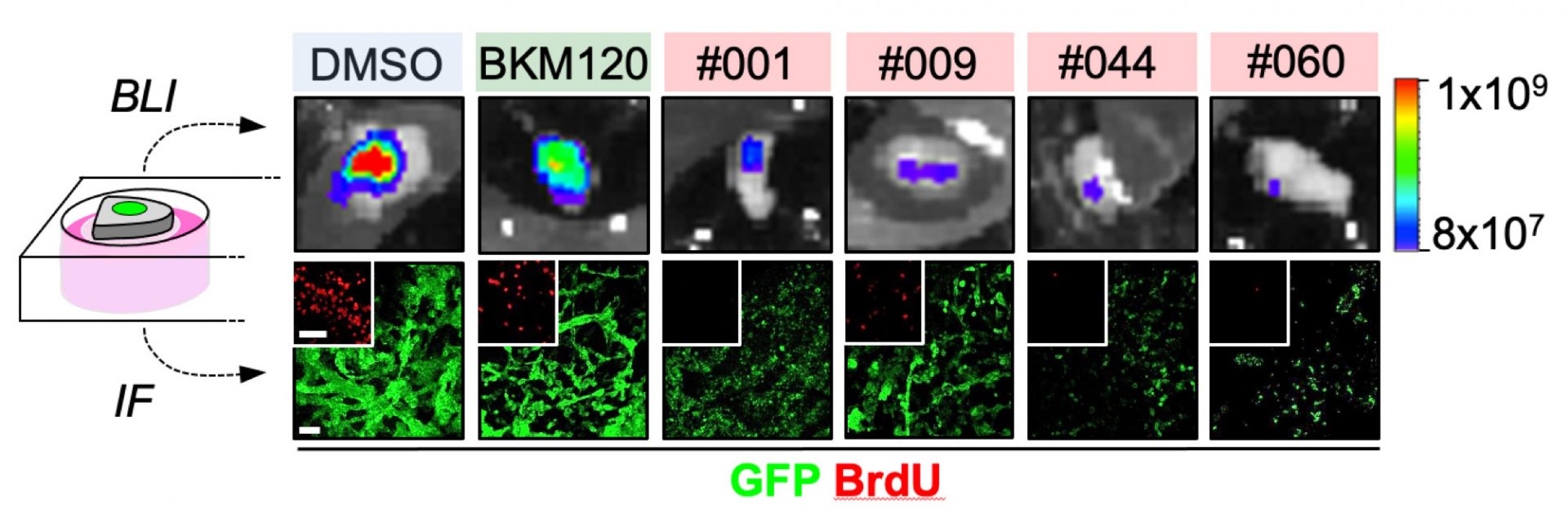
We have created METPlatform, an organotypic culture-based drug screening platform that could evaluate the efficacy of hundreds of compounds to prioritize those targeting metastases while growing in situ. METPlatform is not only applicable to brain metastasis but to almost any organ that could be affected by secondary tumors.
We have extensively validated METPlatform using both FDA-approved and experimental drugs to select hits that were later validated in vivo. Importantly, human samples could be easily incorporated into this drug-screening platform, giving rise to a more comprehensive validation closer to the clinical situation.
Additionally, METPlatform could be characterized with omic approaches to define the response to therapy at the molecular level. By using this approach, we identified mediators of brain metastasis as well as mechanisms of resistance.
We hope that METPlatform will contribute to stop the vicious cycle by which the poor prognosis of patients with brain metastasis is the key argument to justify their exclusion from many clinical trials. We demonstrated that drugs already approved might be effective. Even more, METPlatform has uncovered novel brain metastases biomarkers that could be used to improve the diagnosis and prognosis. Finally, given its potential to be used as a clinically-compatible “avatar”, we envision that this strategy could be incorporated in clinical trials to expand the benefits of personalized therapy by including metastatic disease.