- Resistance
Radiation
- Radiation gets immediate access to the brain but it is not as effective as expected, why?
Stratification of radiosensitive brain metastases based on an actionable S100A9/RAGE resistance mechanism.
Monteiro C, Miarka L, et al. Nature Medicine (2022).
Protocol to generate murine organotypic brain cultures for drug screening and evaluation of anti-metastatic efficacy
Lucía Zhu, Lauritz Miarka, Patricia Baena, María Perea-García, Manuel Valiente. STAR Protocols (2023)
Emerging targets for cancer treatment: S100A9/RAGE
M. Valiente, J.M. Sepúlveda, A. Pérez. ESMO Open (2023)
There are mainly two approaches to provide radiotherapy to the brain. Whole brain radiotherapy (WBRT) and stereotactic radiotherapy (SRS) -a more localized strategy. WBRT is being used less in favour of SRS because the second radiation modality can spare healthy brain, thus avoiding potential long-term toxic effects, and is more effective. However, SRS cannot be always applied to every patient.
We reasoned that it would be interested to know why WBRT is not more effective.
We realized that radiotherapy could be modulated depending on where cancer cells grow and that the brain dramatically increases their resistance to this therapy. Thus, we have performed unbiased approaches to identify molecular changes correlating with increased resistance and found several de-regulated genes that we have validated as potential sources of radioresistant mechanisms.
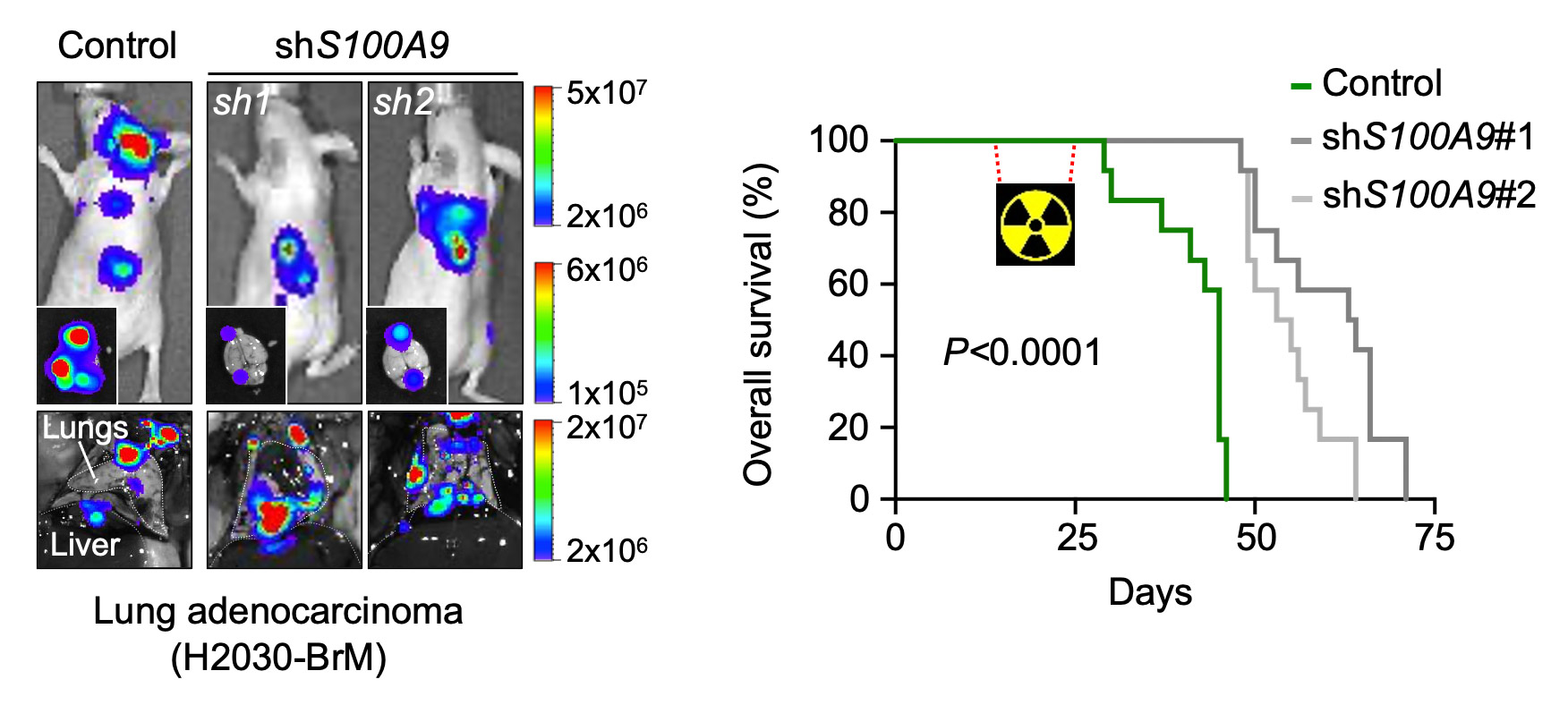
We have identified a key mechanism explaining resistance to radiotherapy. By applying WBRT to our experimental models of brain metastasis we realized that the strong resistance to this therapy was reverted when the expression of S100A9 was silenced in cancer cells. We not only demonstrated that this occurs independently of the source of the primary tumor but that it also applied to patients with radioresistant brain metastases. Indeed, by dissecting the signalling mechanism (S100A9-RAGE-NFkB-JunB) we incorporated inhibitors that reproduced the induction of radiosensitivity obtained with the genetic approach. Furthermore, given the secreted nature of S100A9 in cancer cells, we were able to establish a biomarker compatible with liquid biopsy.
Our findings suggest that it is possible to personalize the use of radiotherapy in patients with brain metastasis based on a biomarker of radioresistance (S100A9) that could be evaluated in liquid biopsy. In addition, we identified potential strategies (RAGE inhibitors) that could be added to radiotherapy to improve its efficacy. It is tempting to speculate that the same resistance mechanism could applied to other brain primary tumors.