- Resistance
Local relapse
- Surgery is a key aspect in the treatment of some patients with brain metastasis. If the metastasis is removed, why is this therapy not curative?
A clinically-compatible drug-screening platform based on organotypic cultures identifies vulnerabilities to prevent and treat brain metastasis.
Zhu L, et al. EMBO Molecular Medicine. (2022).
Protocol to generate murine organotypic brain cultures for drug screening and evaluation of anti-metastatic efficacy
Lucía Zhu, Lauritz Miarka, Patricia Baena, María Perea-García, Manuel Valiente. STAR Protocols (2023)
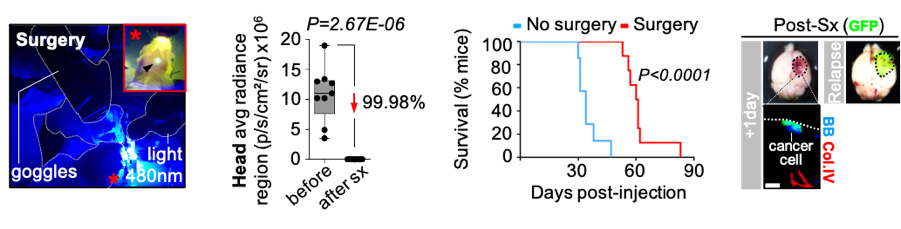
Neurosurgery cannot be always applied since it depends on many aspects (i.e. number of metastasis, location, general status of the patient). However, when applied to eliminate a tumor mass, frequently the metastasis reappears at the same location from which it was excised. This is a key problem that limits the benefits of this local therapy. We thought that studying the process of relapse after surgery was important because its biology was completely unknown.
Since there were no models to study this problem, we created one. An established brain metastasis underwent neurosurgery and then local relapse followed over time using non-invasive techniques such as bioluminescence or other approaches such as histology.
The new model of brain neurosurgery is highly prevalent regarding the relapse phenotype, which can be observed several weeks post-intervention. Transcriptomic analysis shows that differences could be observed with the initial metastasis. However, not all aspects of the biology might be different compared to the original metastasis. For instance, we have identified drugs that target both the original and relapsed metastases effectively.
This new model of brain metastasis represents the possibility to reproduce a very frequent clinical situation for which there are no effective therapeutic strategies. We envision that establishing a molecular map of relapse by studying in a time-lapse manner both the cancer cells left behind, which are the seeds for the relapse, as well as the surgical bed, which represent a particular microenvironment damaged by both the metastasis and the surgery, will allow to establish a therapeutic pipeline for preventing relapse post-surgery.